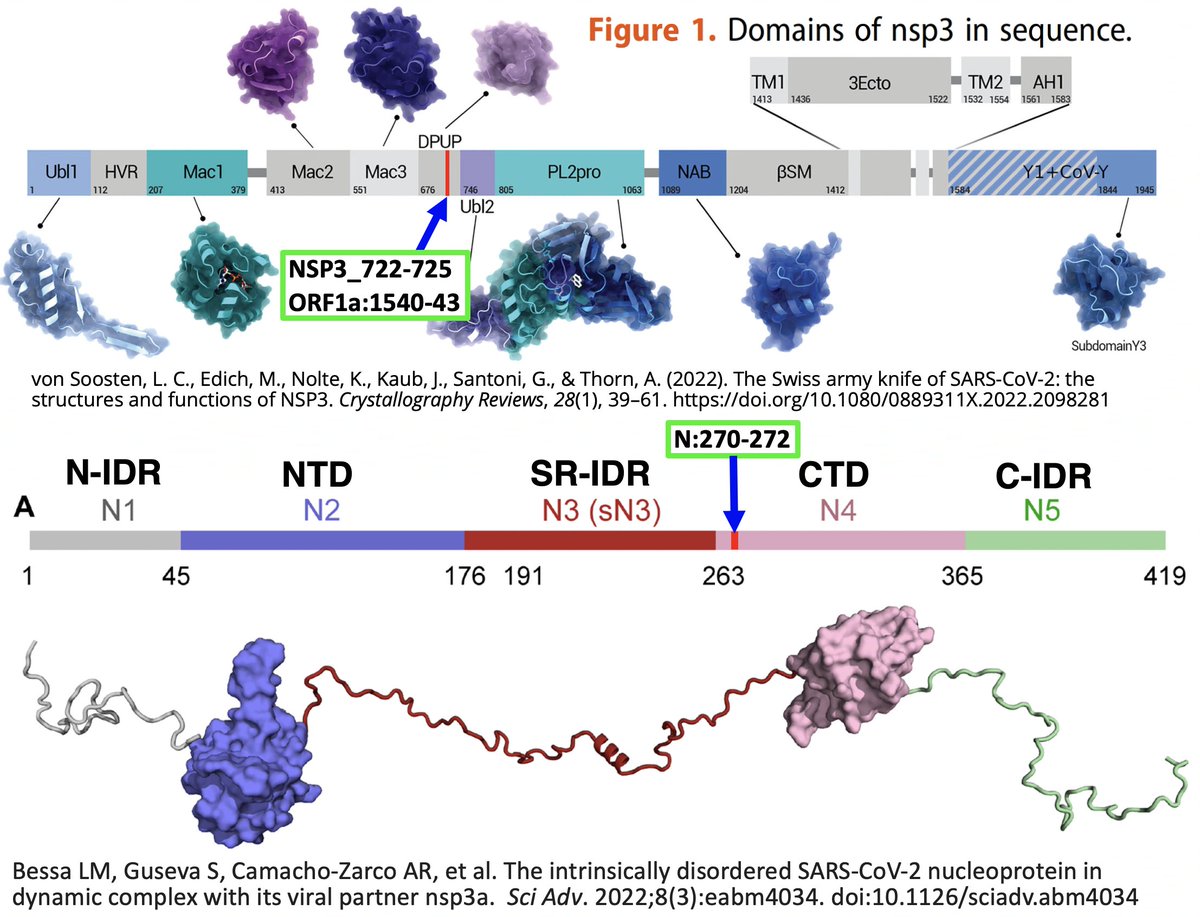
What connects two regions on opposite ends of NSP12, a narrow slice of an obscure NSP3 region (DPUP/SUD-C), & a 3-AA sliver of nucleocapsid (N)? I have no idea, but I’m convinced there’s a link that could help reveal the inner workings of SARS-CoV-2. 1/120
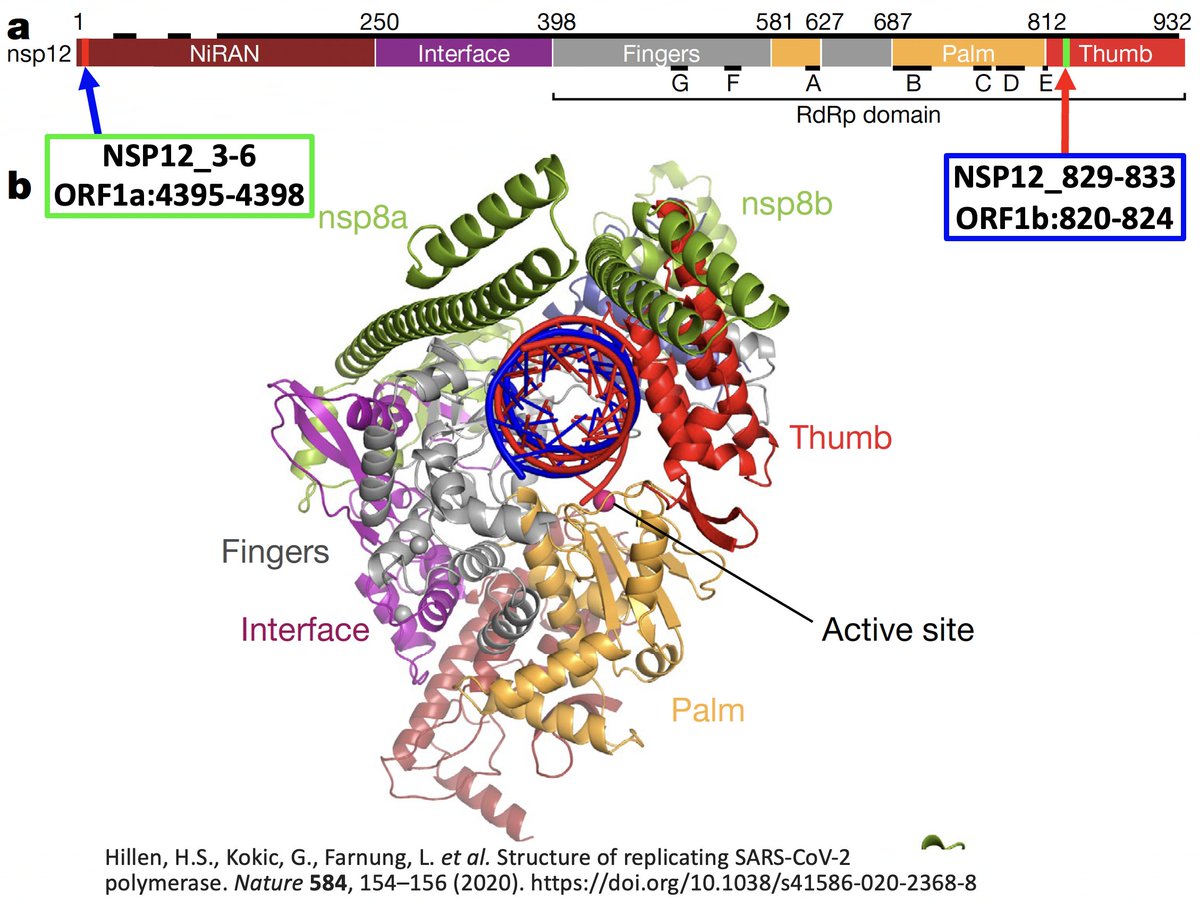
I previously wrote a thread about the strange connection between ORF1a:4395-4398 and ORF1b:820-824 (NSP12_3-6 & NSP12_829-833). There is no known connection between these regions, & they are not close to each other in the NSP12 protein structure. 2/120 x.com/longdeserttrai…
I previously wrote a thread about the strange connection between ORF1a:4395-4398 and ORF1b:820-824 (NSP12_3-6 & NSP12_829-833). There is no known connection between these regions, & they are not close to each other in the NSP12 protein structure. 2/120 x.com/longdeserttrai…
Mutations in both regions are rare, yet they arise in the same sequences again and again, at rates that cannot be coincidental. Furthermore, there have never been any circulating lineages with these paired mutations—they are a chronic-infection specialty. 3/120
(Tweets #4-15 = short detour on chronic infections) Nearly all major SARS-CoV-2 variants have, IMO, originated in chronic infections. And to paraphrase @SolidEvidence, chronic infections are where this virus goes to college. 4/120 x.com/solidevidence/…
(Tweets #4-15 = short detour on chronic infections) Nearly all major SARS-CoV-2 variants have, IMO, originated in chronic infections. And to paraphrase @SolidEvidence, chronic infections are where this virus goes to college. 4/120 x.com/solidevidence/…
Some viruses join fraternities and pick up bad habits (E:T30I, S:L828F), dimming their future prospects, but others emerge transformed, brimming with novel tricks (S:ins16:MPLF, S:L452W, S:∆V483), and ready to take on the world. 5/120
What makes certain human bodies such potent incubators of divergent, world-conquering variants? Much of this remains shrouded in a fog of biological complexity, but one factor is widely acknowledged: the lack of a transmission bottleneck. 6/120
When a virus infects a person it undergoes mutations. Most are harmful and therefore fleeting. But some may evade host immune responses or enable for rapid replication. These mutations start with a single virion but may expand over the course of an infection. 7/120
Given unlimited time, such beneficial mutations would eventually predominate. But transmission, if it occurs, tends to happen quickly, before such mutations have had time to grow. And if transmission only involves a very small number of virions, as with SARS-CoV-2, the… 8/120
…odds of a beneficial mutation transmitting are small; they die in the cradle, asphyxiated by the bottleneck, which grabs the slowly grinding gears of natural selection & spins them like a roulette wheel at every transmission.See @Mahan_Ghafari, @ArisKatzourakis, @pvmarkov 9/120
If multiple mutations must be combined to pull off a fitness-enhancing trick, the odds of them occurring during a normal infection & transmitting become infinitesimal. See excellent paper by @lauring_lab on the subject of bottlenecks. 10/120
But in chronic infections, there is no bottleneck. Evolution has free play. Immune-evasive mutations grow dominant. Diverse variants arise in different organs, then recombine. Unlikely combinations occur & if beneficial, become fixed. The gears of selection grind forward. 11/120
Whether or not mutations that thrive within a host will also prove advantageous in circulation depends on a multitude of factors not entirely understood. Undoubtedly, some mutations that increase intrahost fitness will impede transmission. 12/120